Linus Safety Release
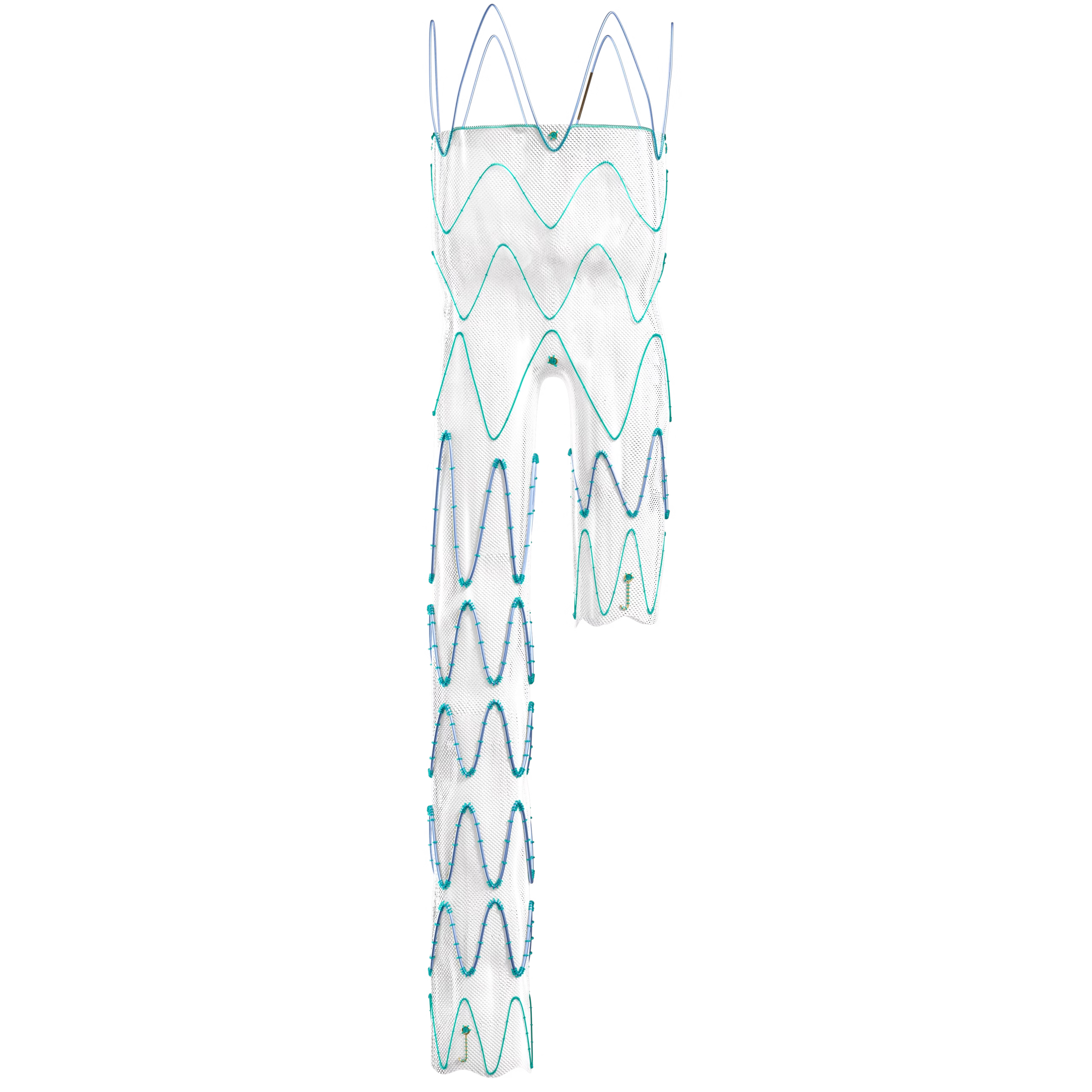
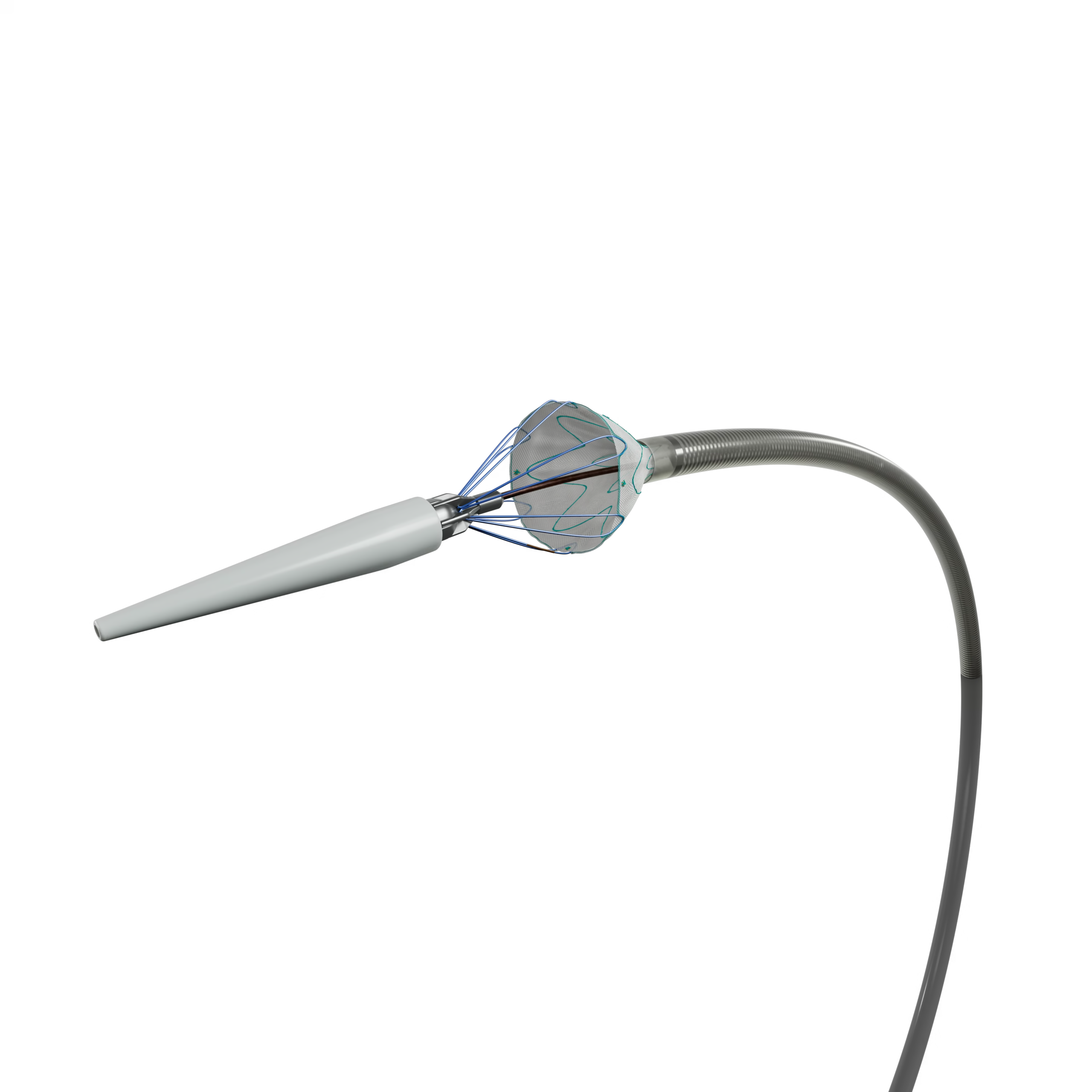
The Linus Safety Release Vascular Stent consists of an intraluminal arterial support prosthesis (stent-graft) and a delivery device. Developed by Braile Biomédica, the product uses a variation of the Z-Gianturco grid, made of Nitinol and coated with polyester fabric. These are self-expanding prostheses with high radial resistance, carefully compressed and inserted into Polyether-Block Amide (PEBA) catheters reinforced with AISI 304V stainless steel and lined internally with Polytetrafluoroethylene (PTFE) and externally with hydrophilic resin. The stent-graft can be fully covered by polyester fabric, with an uncoated proximal stent module that allows for better fixation of the prosthesis, known as "Free-Flow," or with a module partially covered with fabric following the shape of the metallic structure, called "Open-Web." The "Free-Flow" module can have its outer edge protected with a polyester fabric patch.
The release device allows the use of guide wire up to 0.035” (wire not included).
Anvisa Registration Number: 10159030096