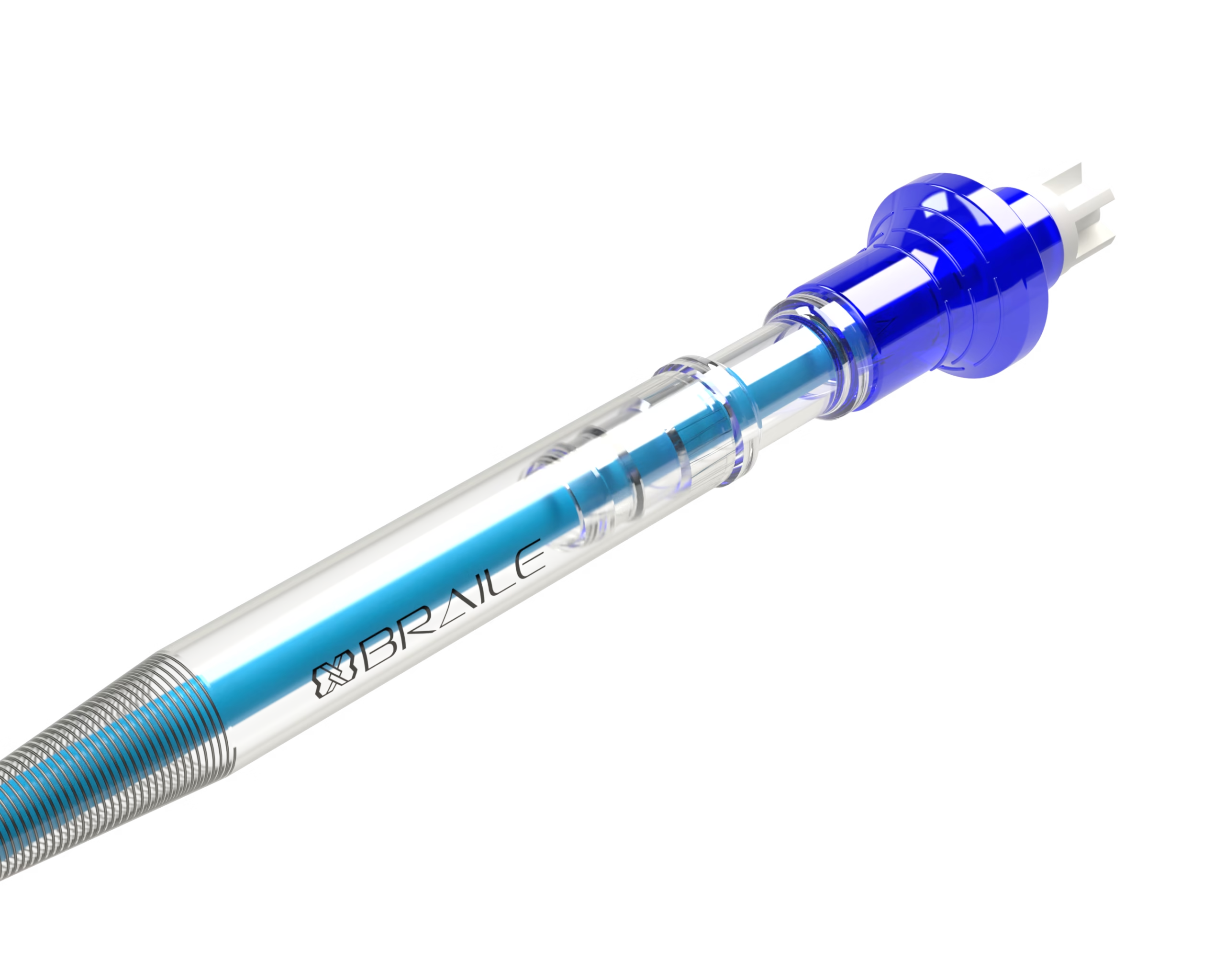
Agile BRCoating® Wired Femoral Venous Cannula
The Agile BRCoating® Femoral Venous Cannula is composed of polyvinyl chloride (PVC), 304 stainless steel, polycarbonate (PC), acrylonitrile butadiene styrene (ABS), 711-8005/USP paint, and BRCoating® coating, composed of recombinant (synthetic) albumin and polyethylene glycol 300 (PEG 300).
The cannula has a wire body with multiple perforation stages, an introducer that allows percutaneous insertion of the cannula and is compatible with a 0.038˝ guidewire, a connector that allows connection to the extracorporeal circulation circuit, and a cap to protect the product before use. The cannula has a BRCoating® coating, composed of recombinant synthetic albumin in association with polyethylene glycol. The cannula has depth markings with ink specifically for medical use.
The Agile BRCoating® Femoral Venous Cannula should be positioned in a blood vessel to drain venous blood into the extracorporeal circuit. Drainage can be achieved by gravity or with the aid of negative pressure in the circuit line.
All components are made of biocompatible and pyrogen-free materials.
Anvisa No.: 10159030117